Contoh Sop Keuangan Perusahaan pdf converter Step 7: All videos related to your search will appear in the page results, Then in the video results choose the video you want to download then click the download button. Step 6: In the search box put the artist name or the title of the video you want to download, After you place the name in the search box then click search. Step 8: In the download page, You can play the video first to find out if the video is appropriate to your needs, To download the video you will see different links and then click the download button, Many video file formats will appear, Now select the format of video you want to download Mp9 8Gp Video, Mp8 Songs. WapSpot is the fastest youtube video downloader site that you can search alot of videos, songs, audio.
Standard operating procedure Title: Management of periodic safety update reports (PSURs) for centrally authorised veterinary medicinal products Status: PUBLIC Lead author Name: Raquel Gopal Signature: on file Date: 28-MAR-12 Approver Name: Kornelia Grein Signature: on file Date: 28-MAR-12 Document no.: SOP/V/4023 Effective date: 30-MAR-12 Review date: 30-MAR-15 Supersedes: SOP/V/4023 (03-JUN-09) TrackWise record no.: 3502 1. Pengertian adobe dreamweaver menurut para ahli. Purpose To enable a transparent procedure for the management of periodic safety update reports (PSURs) for centrally authorised veterinary medicinal products following the granting of a central marketing authorisation by the European Commission and to ensure timely submission and assessment of the PSURs as well as the follow-up of the conclusions and recommendations of the Committee for Medicinal Products for Veterinary Use (CVMP). Scope This standard operating procedure (SOP) applies to CVMP members and alternates, CVMP Pharmacovigilance Working Party members and the European Medicines Agency (EMA) staff of the veterinary medicines sector in the veterinary medicines and product data management unit. Responsibilities It is the responsibility of the Head of Veterinary Medicines to ensure that this SOP is adhered to. The responsibility for the execution of each particular step of this procedure is identified in the right-hand column of section 9 of this SOP. Changes since last revision Consistency updates made in line with the EMA corporate identity and internal organisational restructure. Update of references in Section 6 Related documents.

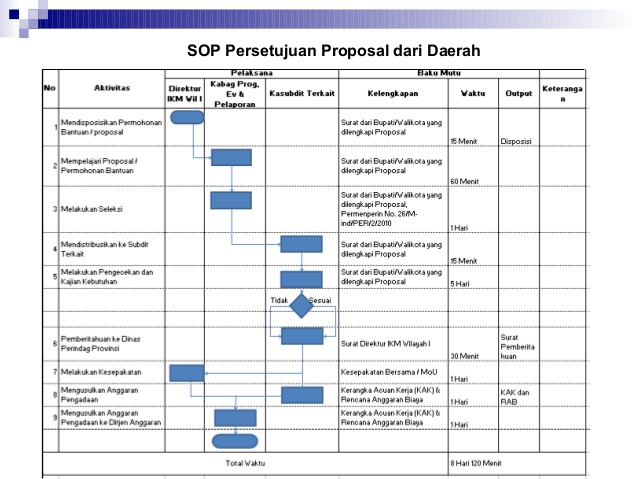
Contoh Sop Prosedur Perusahaan

7 Westferry Circus ● Canary Wharf ● London E14 4HB ● United Kingdom Telephone +44 (0) 0 7418 8400 Facsimile +44 (0)20 7418 8416 E-mail [email protected] Website www.ema.europa.eu An agency of the European Union © European Medicines Agency, 2012. Reproduction is authorised provided the source is acknowledged. Documents needed for this SOP Models as identified by the work instruction for processing of PSURs for centrally authorised products (WIN/V/4028). Related documents Directive 2001/82/EC of the European Parliament and of the Council of 6 November 2001 on the Community code relating to veterinary medicinal products Regulation (EC) No. Majalah maxim. 726/2004 of 31 March 2004 Commission Regulation (EC) No.
